With their PiSCO® Impulse System, our customer Miracor Medical achieved a breakthrough in the treatment of heart attack patients. Mechatronic contributed their share that helped make a difference. We were able to realize the system by testing, optimizing and manufacturing it. Another good example of innovative cooperation in medical engineering.

Portfolio: Miracor Medical
THE DIFFERENCE THAT CAN SAVE YOUR PROJECT
SITUATION
A DEVICE WITHOUT AUTHORIZATION
Sometimes only one piece is missing – but this piece makes all the difference and puts the most important step on hold: authorization.
The device had already been built at Miracor Medical. Yet, it wasn’t ready for series production according to regulations outside Europe. So we took matters in our hands: problem analysis and cause study first, finding a solution second.
Our focus was on the reliability of the device in order to make it fit for series production.
What we did was a thorough testing of all its components, thus improving their technical quality and administering further changes.
Compared to the product development that had already taken place, these were minor adjustments but nonetheless vital for the operation and serviceability of the device.
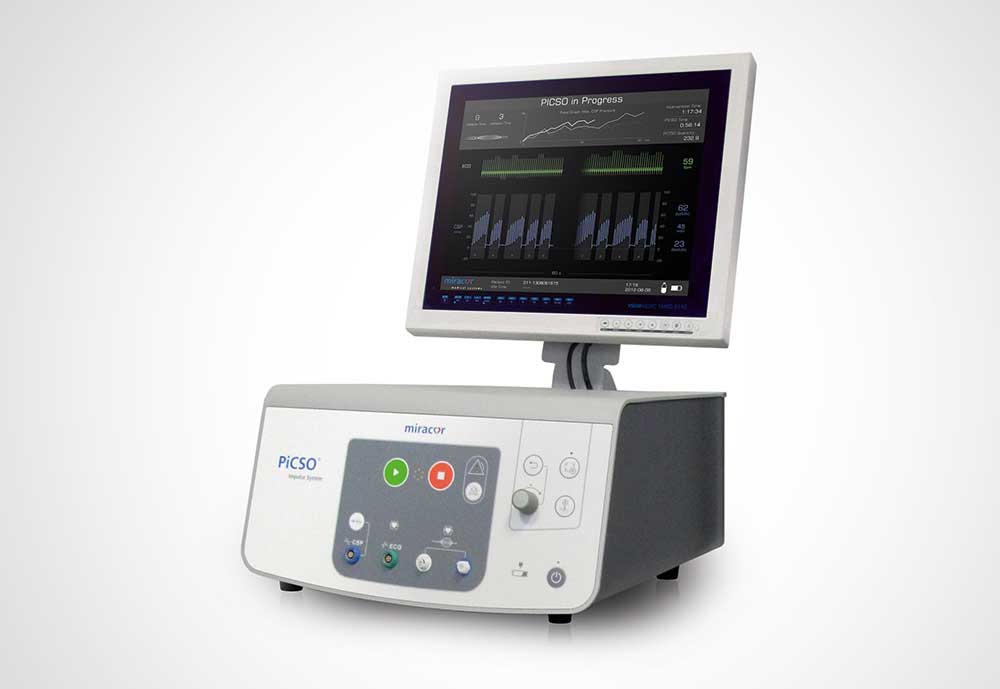
TASK
DOING WHAT NEEDS TO BE DONE
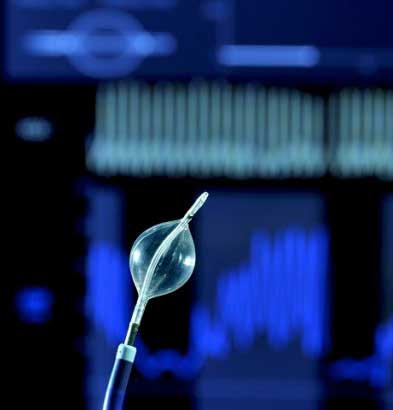
Human lives depend on the functioning of this medical device. For intensive care patients malfunctions must be ruled out, which means: no failure, no faults.
All components have to work reliably: pneumatic blocks, vacuum tanks, pressure tanks, control units as well as pumps, not to mention the electronic parts of the device.
First, it was essential to identify suitable components and integrate them. Then, prototypes were built for the improved consoles. The Mechatronic team established a production line with all the necessary ATEs. Finally, the devices were manufactured and delivered.
The product development that had stalled midway, came to its successful close with the help of Mechatronic.
ACHIEVEMENT
AUTHORIZATION WITH FLYING COLOURS
Our customer Miracor Medical can be proud of their innovative solution for severe heart diseases. The PiSCO® Impulse System is the first coronary sinus treatment method that was introduced to reduce the severeness of the heart attack and to improve micro-circulation in the aftermath of an acute myocardial infarction (AMI).
This technological breakthrough went hand in hand with a breakthrough in realization quality. With the help of Mechatronic, the “best plan ever” became “ready for take-off”. Authorization really means proof of excellence. We like to meet challenges like these. We are precise, reliable and success-oriented.
In the case of Miracor, it was literally an affair of the heart for us.

Miracor Medical SA is a medical device company located in Belgium. They develop innovative solutions for the treatment of severe heart diseases.
SUCCESS
THE IMPACT OF INNOVATION
In the meantime, Miracor Medical has received the label “Breakthrough Device Designation” from the FDA. The American agency for medicine has acknowledged the unique impact of the PiSCO® Impulse System, which means a major innovation in the treatment of AMI patients.
It is not without pride that we look back on our part in this success story and the difference we made in taking this medical device to the level of series production. A vision was completed, a project was saved, and progress was made real. You can trust us with your vital projects because we are specialists. We are looking forward to new challenges.